Permanent farnesylation of lamin A mutants linked to progeria impairs its phosphorylation at serine 22 during interphase | Aging

Phosphorylated Lamin A/C in the Nuclear Interior Binds Active Enhancers Associated with Abnormal Transcription in Progeria - ScienceDirect

A progeria mutation reveals functions for lamin A in nuclear assembly, architecture, and chromosome organization | PNAS

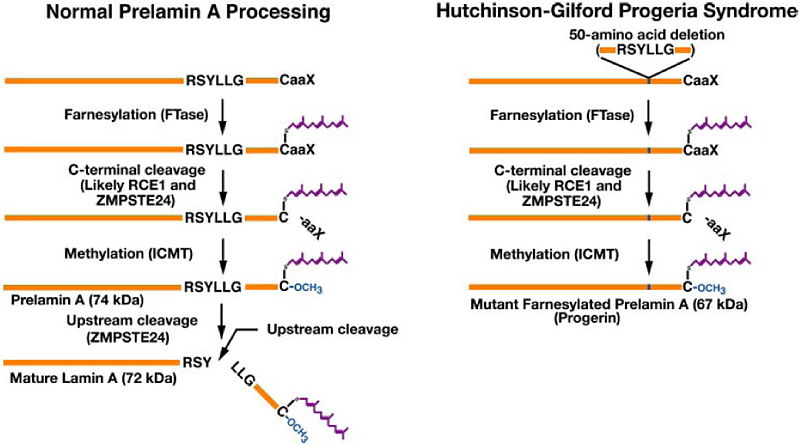
Prelamin A and progerin differ in their processing. A simplified model... | Download Scientific Diagram

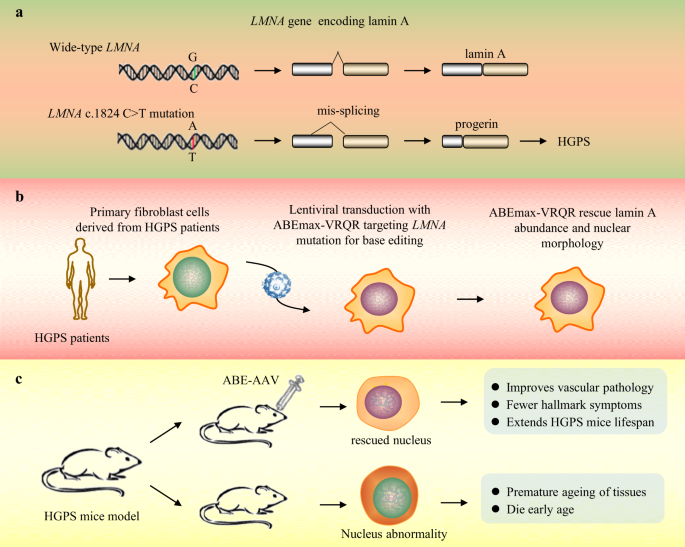
Francis Villatoro on X: "#Nature In vivo base editing rescues Hutchinson–Gilford progeria syndrome in mice (by David R. Liu et al.) https://t.co/YvqGq0aSGg Progeria is typically caused by a dominant-negative C•G-to-T•A mutation (c.1824

Mechanics in human fibroblasts and progeria: Lamin A mutation E145K results in stiffening of nuclei - Apte - 2017 - Journal of Molecular Recognition - Wiley Online Library

Speeding up the clock: The past, present and future of progeria - Swahari - 2016 - Development, Growth & Differentiation - Wiley Online Library

A progeria mutation reveals functions for lamin A in nuclear assembly, architecture, and chromosome organization | PNAS

A progeria mutation reveals functions for lamin A in nuclear assembly, architecture, and chromosome organization | PNAS

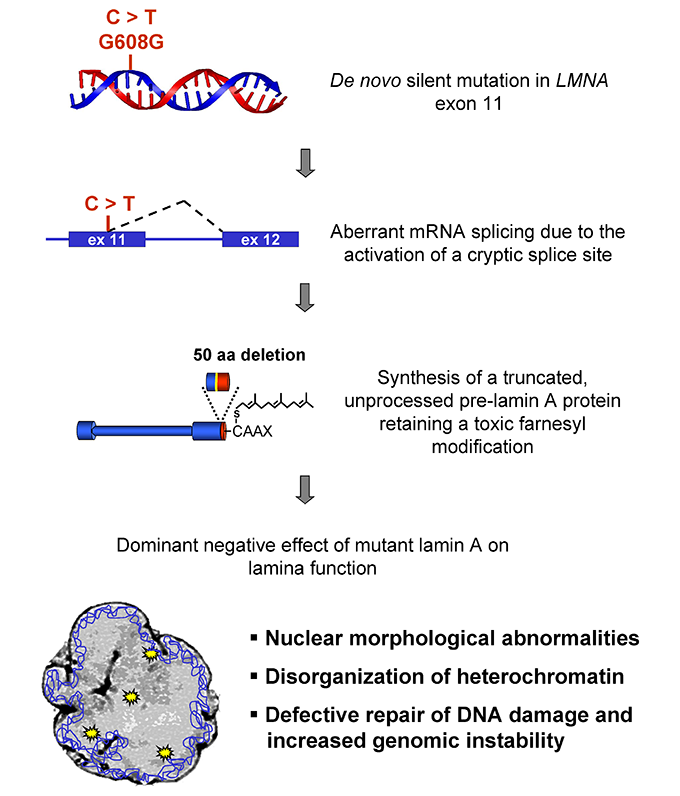
Accumulation of mutant lamin A causes progressive changes in nuclear architecture in Hutchinson–Gilford progeria syndrome | PNAS